Immunoglobulin stimulatory property of aqueous leaf extract of Justicia carnea in male Wistar rats exposed to lead acetate
DOI:
https://doi.org/10.23921/amp.2023v7i1.00070Keywords:
Humoral immunity, Immunoglobulins, Justicia carnea, ToxicityAbstract
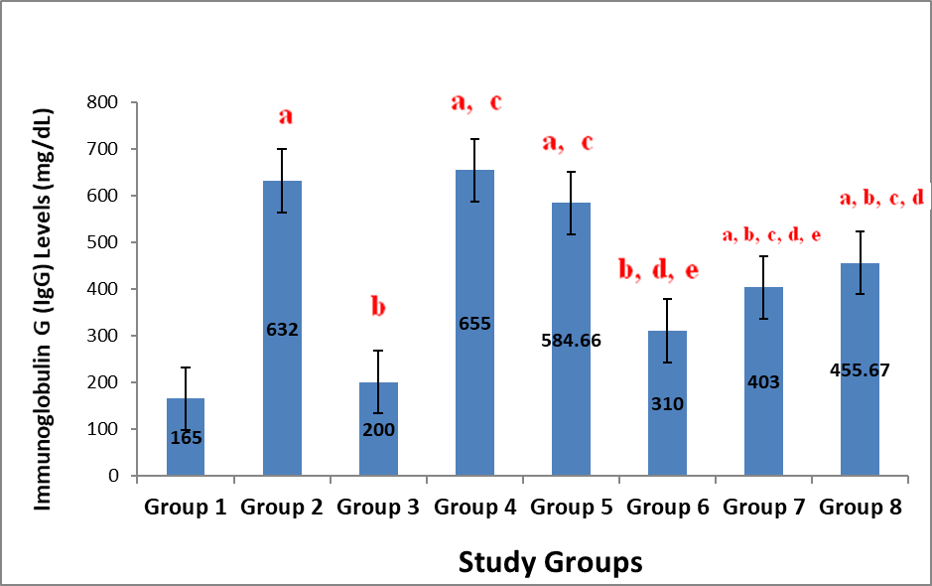
With regards to previously speculated hematinic/hematopoietic potentials of Justicia carnea, the present study set out to evaluate the effect of the administration of the aqueous leaf extract of Justicia carnea on the immunoglobulins levels in two sets of rats (non-lead intoxicated and lead- intoxicated rats). Forty (40) male Wistar rats were obtained and randomly distributed into eight (8) groups of 5 rats each as follows: Group 1 served as negative control/received 1ml of distilled water daily; Group 2 served as positive control/treated with 10mg/kg.bw of lead acetate (Pb); Groups 3 to 5 received 400, 600 and 800mg/kg body weight of aqueous leaf extract of Justicia carnea respectively daily and Groups 6 to 8 received 400, 600 and 800mg/kg bw extract + 10mg/kg.bw Pb respectively daily. At the end of 6 weeks of treatment, blood samples were harvested from the study animals via cardiac puncture after sedating them with 80% chloroform for laboratory analyses. The result of the present study indicated that immunoglobulins (Ig) G, M and A had significant (p<0.05) increases across all extract treated rats. In conclusion, this study has shown that extract possess humoral immunity enhancing potential. However, extrapolating from the present study, the frequency/consumption of the high dose of the extract should be checked in its ethnobotanical application as to prevent any possible consequent undesirable effects of autoimmune conditions.
Downloads
Metrics
References
Safiri S, Kolahi AA, Noori M, Nejadghaderi SA, Karamzad N, Bragazzi NL, Sullman MJM, Abdollahi M, Collins GS, Kaufman JS, Grieger JA. Burden of anemia and its underlying causes in 204 countries and territories, 1990-2019: results from the Global Burden of Disease Study 2019. J Hematol Oncol. 2021 Nov 4;14(1):185.
GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet. 2020 Oct 17;396(10258):1204-1222.
Thachil J, Owusu-Ofori S, Bates I. Haematological Diseases in the Tropics. In: Farrar J, Hotez PJ, Junghanss T, Kang G, Lalloo D, White NJ, editors. Manson’s Tropical Infectious Diseases. Chapter 65, 2014;894-932.e7.
James PB, Wardle J, Steel A, Adams J. Traditional, complementary and alternative medicine use in Sub-Saharan Africa: a systematic review. BMJ Glob Health. 2018 Oct 31;3(5):e000895.
Njume C, Goduka NI. Treatment of diarrhoea in rural African communities: an overview of measures to maximise the medicinal potentials of indigenous plants. Int J Environ Res Public Health. 2012 Oct 26;9(11):3911-33.
Okaiyeto K, Oguntibeju OO. African Herbal Medicines: Adverse Effects and Cytotoxic Potentials with Different Therapeutic Applications. Int J Environ Res Public Health. 2021 Jun 2;18(11):5988.
Onyeabo C, Achi NK, Ekeleme-Egedigwe CA, Ebere CU, Okoro CK. Haematological and biochemical studies on Justicia carnea leaves extract in phenylhydrazine induced-anemia in albino rats. Acta Sci Pol Technol Aliment. 2017 Apr-Jun;16(2):217-230.
Anthonia OC, Ikechukwu UR, Uzoma NO, Uchenna Sunday EL. Nutritive properties of aqueous extract Justicia carnea leaves and its effects on haematological and some biochemical indices of anaemia induced male wistar albino rats. Biomedical Research. 2019;30(4).
Faluyi O. Nigeria’s medicinal plant: Justicia carne (Ewe eje). Punch 2021 Jun 13. Available from: https://punchng.com/nigerias-medicinal-plant-justicia-carne-ewe-eje/ [Accessed online in January, 2023]
Minaopunyea O-OB, Chimeziea OH. Ameliorative potentials of Justicia Carnea and Cnidoscolus Aconitifolius on the fecundity of chloramphenicol-induced lymphoma rats. Journal La Lifesci. 2022 Mar 30;3(2):65-75.
Imohiosen O. Phytochemical analysis on aqueous leaf extract of Justicia carnea (Acanthaceae) and its antibacterial activity on some isolated bacterial. American Journal of Food Science and Technology. 2023 May 7;2(1):16-20.
Akintimehin ES, Karigidi KO, Omogunwa TS, Adetuyi FO. Safety assessment of oral administration of ethanol extract of Justicia carnea leaf in healthy wistar rats: hematology, antioxidative and histology studies. Clinical Phytoscience. 2021 Jan 3;7(1).
Ani ON, Udedi SC, Asogwa KK, Enemali MO, Onwelumadu CM, Ikedife KS. Inhibitory potential and antidiabetic activity of leaf extracts of Justicia carnea. International Journal of Biochemistry Research & Review. 2020 Jun 30;34–45.
Ukpabi-Ugo JC, Ndukwe PA, Iwuoha AG. Hepatoprotective effect of methanol extract of Justicia carnea leaves on carbon tetrachloride-intoxicated albino rats. Biochemistry & Analytical Biochemistry. 2019;8(381):2161-1009.
Tawaha K, Alali FQ, Gharaibeh M, Mohammad M, El-Elimat T. Antioxidant activity and total phenolic content of selected Jordanian plant species. Food Chemistry. 2007;104(4):1372-1378.
Wood J, Yasmin-Karim S, Moreau M, Kumar R, Akwanwi J, Derek A, Atoneche F, Kress J, Ngwa AW. Characterization of isolated extracts from Justicia plant leaves used as remedy for anemia. Molecules. 2020 Jan 25;25(3):534.
Ajuru MG, Kpekot KA, Robinson GE, Amutadi MC. Proximate and Phytochemical Analysis of the Leaves of Justicia carnea Lindi. and Justicia secunda Vahl and its Taxonomic Implications. Journal of Biomedicine and Biosensors. 2022 Dec 15;2(1):1-12.
Okocha BI, Orie KJ, Duru RU, Ngochindo RL. Analysis of the active metabolites of ethanol and ethyl acetate extract of Justicia carnea. African Journal of Biomedical Research. 2023 Jul 14;26(1):109-17.
National Research Council [NRC]. Guidelines of the National Institute of Health (NIH) for care and use of laboratory/research animals. 2011. Available from: https://www.ncbi.nlm.nih.gov/books/NBK54050. (Accessed January, 2023)
Sankeshwari RM, Ankola AV, Bhat K, Hullatti K. Soxhlet versus cold maceration: Which method gives better antimicrobial activity to licorice extract against Streptococcus mutans? Journal of the Scientific Society. 2018 May-Aug;45(2):67-71.
Amah-Tariah FS, Bekinbo MT, Dapper DV. Comparative study of serum immunoglobulin levels in healthy pregnant and pregnant subjects with HIV and malaria infection in Port Harcourt, Nigeria. International Research Journal of Medical Sciences. 2016 Sep; 4(9):11-16.
Fruman DA, Cantley LC. Idelalisib--a PI3Kδ inhibitor for B-cell cancers. N Engl J Med. 2014 Mar 13;370(11):1061-1062.
Pieper K, Grimbacher B, Eibel H. B-cell biology and development. J Allergy Clin Immunol. 2013 Apr;131(4):959-971.
Goldberg BS, Ackerman ME. Antibody-mediated complement activation in pathology and protection. Immunol Cell Biol. 2020 Apr;98(4):305-317.
Chaplin DD. Overview of the immune response. J Allergy Clin Immunol. 2010 Feb;125(2 Suppl 2):S3-23.
Chandra RK, Newberne PM. Interactions of Nutrition, Infection, and Immune Response in Animals. In: Nutrition, Immunity, and Infection. Chapter 7. New York: Plenum Press. 1977.
Greensmith J, Whitbrook A, Aickelin U. Artificial Immune Systems. In: Gendreau M, Potvin JY (eds). Handbook of Metaheuristics. Chapter 14. Boston, MA: Springer. 2010.
Yamada K, Hung P, Park TK, Park PJ, Lim BO. A comparison of the immunostimulatory effects of the medicinal herbs Echinacea, Ashwagandha and Brahmi. J Ethnopharmacol. 2011 Sep 1;137(1):231-235.
Khan SR, van der Burgh AC, Peeters RP, van Hagen PM, Dalm VASH, Chaker L. Determinants of Serum Immunoglobulin Levels: A Systematic Review and Meta-Analysis. Front Immunol. 2021 Apr 7;12:664526.
Draborg A, Izarzugaza JM, Houen G. How compelling are the data for Epstein-Barr virus being a trigger for systemic lupus and other autoimmune diseases? Curr Opin Rheumatol. 2016 Jul;28(4):398-404.
Rai T, Wu X, Shen B. Frequency and risk factors of low immunoglobulin levels in patients with inflammatory bowel disease. Gastroenterol Rep (Oxf). 2015 May;3(2):115-121.
Published
How to Cite
License
Copyright (c) 2023 Quench Academy of Medical Education and Research (QAMER)

This work is licensed under a Creative Commons Attribution 4.0 International License.
The authors warrants and represents that the submitted MANUSCRIPT is an original work and has not been published before in any form, and that it does not infringe upon any copyright or other right(s), that it does not contain infringing, libelous, obscene or other unlawful matter, that he/she is the sole and exclusive owner of the rights here-in conveyed to the Publisher, and that he/she has obtained the customary permission from the copyright owner or his legal representative whenever a text/passage from copyrighted material is quoted or a table or illustration from such material is used. The Author(s) will indemnify the Publisher for, and hold the Publisher harmless from any loss, expense or damage occasioned by any claim or suit by a third party for copyright infringement or arising out of any breach of the foregoing warranties as a result of publication of the Article. The Article shall be delivered to the Publisher free of copyright charges. In the event that the Article is not accepted and published by Publisher, this agreement becomes null and void.
Sherpa/Romeo publisher policy can be viewed at Annals of Medical Physiology - Sherpa/Romeo Policy
Plum X metrics
Article level metrics are shown here









